 Social Cognition in Ravens
Social Cognition in Ravens
by Thomas Bugnyar,
University of Vienna, Austria
Reading Options:
Abstract
Complex social life has been proposed as one of the main driving forces for the evolution of higher cognitive abilities in humans and non-human animals. Until recently, this theory has been tested mainly on mammals/primates, whereas little attention has been paid to birds. Indeed, birds provide a challenge to the theory, on one hand because they show high flexibility in group formation and composition, on the other hand because monogamous breeding pairs are the main unit of social structure in many species. Here I illustrate that non-breeding ravens Corvus corax engage in sophisticated social interactions during foraging and conflict management. While Machiavellian-type skills are found in competition for hidden food, the formation and use of valuable relationships (social bonds) seem to be key in dealing with others in daily life. I thus argue that ravens represent a promising case for testing the idea that sophisticated social cognition may evolve in systems with a given degree of social complexity, independently of phylogeny.
Keywords: food caching and pilfering, perspective taking, conflict management, social bond, ravens, Corvus corax
Acknowledgements: This work was supported by the Austrian Science Fund (FWF; grants J2064, J2225; R31-B03, Y366-B17) and the European Science Foundation (ESF-EUROCORES framework TECT: I105- G11). Permanent support has been provided by the ‘Verein d. Förderer KLF’ and the Herzog von Cumberland Stiftung. I would like to thank the editor and three anonymous reviewers for their constructive comments on a previous version of the paper. Contact information: Thomas Bugnyar, Department of Cognitive Biology, University of Vienna, Althanstrasse 14, 1090 Wien, Austria; email: thomas.bugnyar@univie.ac.at
Introduction: Why study social cognition in ‘moderately social’ birds?
Aside from problems related to foraging (Milton, 1988; Parker and Gibson, 1977), life in individualized social groups has been considered as one of the main driving forces for brain evolution (Jolly, 1966; Humphrey, 1976; Whiten and Byrne, 1988). Support for this ‘social brain’ hypothesis comes from studies correlating relative brain size with group size in non-human primates and in some other mammalian taxa (e.g. Dunbar, 1992; Dunbar, 1998; Pérez-Barbería, Shultz and Dunbar, 2007). In addition, field studies on primates and horses reveal effects of social competence on offspring survival (Silk, Alberts and Altmann, 2003; Cameron, Setsaas and Linklater, 2009) and some comparative studies in birds show that social species tend to outperform less social species in experimental tasks (e.g. Bond, Kamil and Balda, 2003).
Despite these encouraging findings, many open questions remain, notably about what constitutes social complexity and what type of cognition is selected for (e.g. Cheney and Seyfarth, 1990, 2007; Barrett, Henzi and Dunbar, 2003; Moll and Tomasello, 2007; Bond, Wei and Kamil, 2010). In birds and some mammals, for instance, brain size does not correlate with group size but with long-term partnerships (Dunbar and Shultz, 2007), indicating that dealing with a particular individual over time, rather than dealing with many individuals, may be cognitively challenging (see also Emery, Seed, von Bayern and Clayton, 2007). Furthermore, in a number of mammalian and avian species, groups are not cohesive units but frequently change in respect to size and composition, with individuals joining, leaving and re-joining (parts of) the group later (Tyack and de Waal, 2003; Emery, 2006). It has been proposed that such systems with high degree of fission-fusion dynamics are particularly challenging in cognitive terms, since individuals have to cope with the temporal absence of others, making it difficult to update information and knowledge about others (Aureli et al., 2008). Yet, empirical examples are focused on mammals such as primates (Amici, Aureli and Call, 2008), cetaceans (Mann, Connor, Tyack and Whitehead, 2000), carnivores (Holekamp, Sakai and Lundrigan, 2007), and elephants (McComb, Moss, Durant, Baker and Sayialek 2000; McComb, Moss, Sayialek and Baker, 2001).
In this paper I review recent studies on the common raven Corvus corax, a large-brained songbird renowned for its high ecological flexibility and scavenging foraging style (Ratcliffe, 1997). Compared with other corvids, it is usually characterized as a moderate social species, since adults defend breeding territories and are usually found in pairs (Boarman and Heinrich, 1999). However, those birds that do not have a territory – representing the class of non-breeders, i.e. young immature birds and sexual mature birds without a partner – tend to form groups that can be characterized by high fission-fusion dynamics (Heinrich, 1988; Marzluff and Angell, 2005). Group formation is advantageous in finding and accessing large but unpredictable food sources like carcasses and kills (Heinrich and Marzluff, 1991; Marzluff and Heinrich, 1991; Bugnyar and Kotrschal, 2002a). Probably because of benefits associated with foraging, non-breeder groups tend to be joined by adult pairs outside the breeding season and/or the year round, when no breeding territories are available (Braun, Walsdorff, Fraser and Bugnyar, 2012). Focusing on two aspects of social life, competition over food and the use of affiliate relations or social bonds, I argue (i) that the social system of ravens is more complex than previously thought and (ii) that this bird species represents a promising case for testing the idea that sophisticated social strategies and cognitive skills may evolve in systems with a given degree of social complexity, independently of phylogeny.
Social aspects of food caching
Food for thought.
Imagine a flock of ravens that have just found a dead moose: not all birds manage to feed because dominants try to monopolize those parts of the carcass that are not covered by skin and allow them access to the meat. One of those dominants flies off after a few minutes with its throat pouch filled with meat; another raven from the crowd flies off in same direction, but without any food. The raven with food does not go far but lands behind a rock in only a few hundred meters distance to the carcass. There, it puts down its piece of meat, sticks it into a crevice and covers the upper part with surrounding debris; it then stretches and turns its head, apparently visually scanning the area, before it leaves the cache and flies back to the carcass. As soon as it has left, another raven flies out of dense trees nearby, making a bee-line for the cache – it is the same bird that has left the carcass without food immediately after the dominant bird. Suddenly, a third raven appears – without food – from the direction of the food source. The raven approaching the cache immediately changes its direction and starts digging in the soil about 10m far from the actual cache. The newcomer lands there, displaces the digging ravens and starts digging, too; however, it soon gives up and flies back to the carcass. The remaining raven now flies directly to the cache, recovers the food and leaves with it in another direction.
Such a scenario is typical for group foraging ravens. When faced with competition for a large food source, they hardly eat but carry off consecutive loads of food for scatter hoarding at a moderate distance to the feeding site (Heinrich and Pepper, 1998). Other ravens may try to find and pilfer those caches, apparently taking into account the behavior of the cacher as well as the behavior of other potential pilferers. The competition for hidden food thus results in seemingly sophisticated maneuvers and counter-maneuvers from both, cachers and pilferers (Bugnyar and Kotrschal, 2002b).
Competition over hidden food
Caching of food for later consumption is a behavioral trait found in most corvids (ravens, crows, magpies and jays; de Kort, Tebbich, Dally, Emery and Clayton, 2006). As short-term cachers, ravens typically recover the food within a few hours up to a few days after the caches have been made. Notably, they tend to return to their caches when the other ravens have left the scene (Heinrich and Pepper, 1998). For group-foraging ravens, food caching may thus primarily reflect a strategy to secure food from conspecifics, rather than a strategy to save surplus food for later use. This stands in contrast to reports from some other corvids like Pinyon jays Gymnorhinus cyanocephalus, which routinely make caching trips in groups (Marzluff and Balda, 1992).
Competition for cached food, however, has been described for a number of corvids and appears to be based on memory for observed caches (Bednekoff and Balda, 1996a,b; Emery and Clayton, 2001). Experiments on captive ravens reveal that success in finding others’ caches is directly linked to the opportunity of observing them being made (Heinrich and Pepper, 1998; Bugnyar and Kotrschal, 2002b), whereas olfactory cues play no or only a minor role in locating hidden food (compare Harriman and Berger, 1986). This is corroborated by the fact that ravens have little problems in remembering up to 25 observed caches (Braun and Bugnyar, unpubl. data) and their memory for observed caches seems excellent for at least 24 hours (Heinrich and Pepper, 1998). Given that pilferers need to observe others making caches to be able to learn about the exact locations, pilfering is rarely a by-product of caching activities. Instead, ravens face the decision whether to try to get food directly from the source or to follow others leaving for caching to get a chance for pilfering (as described in the example above). Their choices can be modeled as a producer-scrounger game (Giraldeau and Caraco, 2000), with individuals caching food they got from the carcass acting as producers and those pilfering caches from others acting as scroungers. Importantly, the roles of individual birds are not fixed but may change flexibly between rounds, e.g. from being the producer/cacher at one occasion to being the scrounger/pilferer at another occasion (Bugnyar and Kotrschal, 2002a,b).
Clearly, pilfering imposes high costs to cachers, as any benefit of storing food is diminished when the food gets stolen before it can be recovered (Andersson and Krebs, 1978). One possibility to deal with the problem is to reduce the risk of being observed (see Vander Waal and Jenkins, 2003, for alternatives). Indeed, ravens engage in several countertactics such as increasing the distance to conspecifics for caching and using obstacles to hide from view (Bugnyar and Kotrschal, 2002b). They may also come back and quickly retrieve and/or aggressively defend their caches when other ravens come close. Interestingly, they do this specifically with birds that have been in the vicinity at the time of caching, whereas they refrain from going back to their caches with birds that came later (Bugnyar and Heinrich, 2005). Similar skills have been described for Western scrub jays Aphelocoma californica (Emery and Clayton, 2001; Dally, Emery and Clayton, 2005, 2006) and Clark’s nutcrackers Nucifraga columbiana (Clary and Kelly, 2011), indicating that paying attention to others at caching is key for proper cache protection in corvids.
Interestingly, similar arguments can be put forward for birds acting as pilferers. On one hand, ravens behave inconspicuously: they observe others caching from a distance and wait for the cachers to leave before they start a pilfering attempt (Bugnyar and Kotrschal, 2002b). This suggests that they are capable of controlling their intention and do not approach the desired food immediately, effectively avoiding any cache defense by the storer. On the other hand, they may also pay attention to other ravens in their vicinity and adjust their timing of pilfering depending on whether or not those potential competitors were present at caching. Specifically, they quickly attempt pilfering when others were around before but refrain from approaching the cache when others came later (Bugnyar and Heinrich, 2005). Finally, if caught in act by the cacher or a (dominant) competitor, pilferers may engage in displacement behaviors like digging in the soil or manipulating objects (Heinrich, 1999; Bugnyar and Heinrich, 2006), which may function to lead others away from the actual cache (Bugnyar and Kotrschal, 2004). To my knowledge, surprisingly little is known about the behavior of pilferers in corvids other than ravens. Aside of some anecdotal reports (e.g. Clayton & Emery, 2004; Källander, 2007), I am not aware of any experimental studies.
Does competition over hidden food select for advanced cognition? Under naturalistic set-ups, both cachers and pilferers engage in behavioral maneuvers that function to deceive others, i.e. they conceal information (e.g. by hiding outside view) and provide false information (e.g. by distracting others from the cache location). Cognitively, such deceptive maneuvers may be based on advanced skills like episodic-like memory for particular individuals (‘who has been around when and where’; Dally, Emery and Clayton, 2006) and, possibly, even perspective taking and knowledge attribution (‘that particular individual has or has not seen the cache and thus is knowledgeable or ignorant about its location’; Bugnyar and Heinrich, 2005; Clayton, Dally and Emery, 2007). Alternatively, these behaviors could be interpreted as orienting on behavioral cues and/or following a combination of learned rules (‘that particular individual was present at time of caching and thus likely pilfers caches’; e.g. Penn and Povinelli, 2007).
In the last few years, I conducted a series of experiments to distinguish between these interpretations. Specifically, I focused on the ravens’ knowledge about others when they act as pilferers (Table 1). This allowed me to use a human experimenter as cacher and thereby effectively control variables such as the number and location of caches and the exact use of cover material in the experiments (Table 2). Test subjects were always bystanders at two caching events which, after some delay, were confronted with competitors with the same, less, or no information about the caches (achieved by being also a bystander at both caches, one of the caches, or none of the caches; Figure 1a,b). Results revealed that birds could instantly (first trial) differentiate between competitors: they quickly approached either of the caches when confronted with conspecifics that were informed about both locations, whereas they selectively chose which cache to pilfer first when confronted with competitors that could see only one of the caches being made. Note that the focal subject always got a head start and the cover material was always placed in a way that all but the focal subject could see the food in the caches at the time of testing (Figure 1a,b), rendering the possibility of behavioral or emotional cueing from competitors unlikely (Table 2). These findings support the hypothesis that ravens can remember who was visually present at which caching events and that they can relate the presence at caching with a high risk of subsequent pilfering (Bugnyar, 2011).
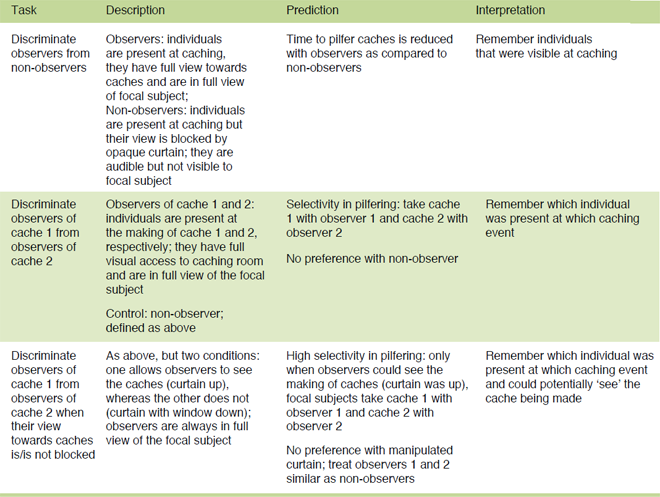
Table 1. Overview of the studies designed to test for the ravens’ ability to differentiate between competitors with different visual experience at caching. The experiments follow a similar procedure but increase in complexity.
In a next step, I tried to disentangle whether ravens base their pilfer decisions on their own perspective or, to some extent, also on the other’s perspective. Now, bystanders were trained to sit on a perch behind an opaque curtain that was either intact or partially intact, i.e. a window was cut out in the upper part at the height of the perch. In the intact condition, the curtain prevented the subject from watching the caching but also other bystanders from seeing their competitor; in the partially intact condition, the curtain still prevented the subject from seeing the caches being made but the view of other bystanders was not affected, i.e. they could see the competitor (Figure 2a). In a third condition, the curtain was pulled up so that the subject could watch the caching and bystanders could see the competitor. As in the previous experiment, two caches were made by a human experimenter and the tested raven got a head start in pilfering the caches in the presence of one of two possible competitors, observer of cache 1 or observer of cache 2 (Table 1, 2). Note that the curtain was always pulled up and competitor positioned on the ground during testing (Figure 2b). As in the previous study, the ravens instantly (first trial) matched the caches to the competitors, i.e. they pilfered cache 1 with observer of cache 1 and cache 2 with observer of cache 2. Interestingly, they did so only when the curtain was up at caching, i.e. the other raven could actually see the cache being made. In the partially intact curtain condition, in contrast, they did not show any preference for pilfering a particular cache first, performing similar to the intact curtain condition. Critically, in the partially intact condition the test subject could see the competitor at caching but the competitor’s view towards the cache was blocked. This supports the assumption that ravens base their choices not only on the memory of their own perspective (‘whom they could see at caching’) but they seemingly also take aspects of the other’s perspective into account (‘what the other could see at caching’).

Table 2. Overview of the control procedures implemented in the described studies.
Although the result of the last experiment is in line with the argument that maneuvers at food caching/pilfering can be explained with sophisticated cognition, it does not require full attribution skills, i.e. a Theory of Mind in the human sense (compare Povinelli and Vonk, 2003; Penn and Povinelli, 2007). For instance, the birds could have tracked the other’s line of sight behind the curtain and, on this basis, inferred their ‘knowledge state’ and risk of pilfering (Bugnyar, 2011). Indeed, experiments on gaze following showed that ravens are skilled in following other’s line of sight behind visual barriers (Bugnyar, Stöwe and Heinrich, 2004; Schloegl, Kotrschal and Bugnyar, 2007). Even more parsimonious would be the explanation that birds could have used subtle behavioral cues like head or body orientation to judge the other’s subsequent behavior. Although we attempted to control for such cues as much as possible at testing (i.e. by making both food pieces visible to the competitor; Table 2), this interpretation cannot be fully ruled out for the observation phase at caching. However, we explicitly tested for the use of gaze direction (defined as head and eye orientation) in another series of experiments involving the same individuals. Results of those studies were clear-cut: ravens naïve about a cache location were not able to learn to use gaze cues given by a conspecific and a human experimenter, respectively, in > 100 trials (Schloegl, Kotrschal and Bugnyar, 2008).
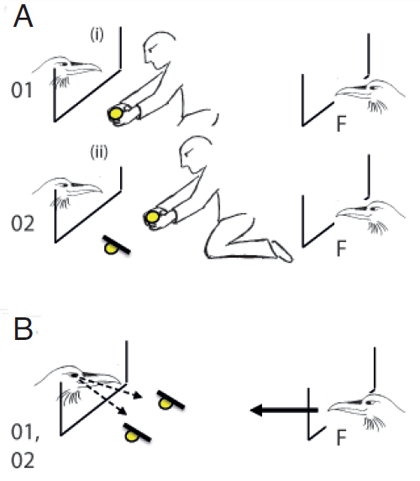
Figure 1. Sketch of the experimental set-up A. during caching and B. during testing. A. Two ravens can observe a human experimenter making caches. Whereas the focal subject (F) is present at both caching events (i, ii), the identity of potential competitors switches between caching events (O1 = observer of cache 1; O2 = observer of cache 2). B. At testing, one of the two potential competitors (O1 or O2) is present and the focal subject (F) gets a head start for pilfering (symbolized by arrow with full line). Note that the skewed positioning of the covers on the caches allows the competitor to see either of the ‘hidden’ food pieces (symbolized by arrows with broken lines).
Finally, the ravens’ excellent performance in the pilfer setup cannot be explained by simple emotional and behavioral rules, which have recently been used successfully to model some cache protection skills (van der Vaart, Verbrugge and Hemelrijk, 2012). So far, these models cannot distinguish between different observers (‘who has watched when’). Moreover, they are based on the assumption that recovery rates of cachers are mediated by their arousal state, which in turn is affected by being observed. Unfortunately, nothing is known about the arousal state of bystanders that simply watch other bystanders at caching as was the case in the current experiments focusing on pilferers.
Taken together, ravens instantly differentiate between competitors with different knowledge states about food caches in several experiments. They likely remember who was present at which caching event and choose their behavioral strategies accordingly. Moreover, pilferers seem to be able to judge the other’s perspective by taking details of the other’s visual behavior into account, like its line of sight and its position relative to optical barriers. These findings support the assumption that maneuvers seen at pilfering are based on advanced socio-cognitive skills and may represent the first building blocks of abilities that go together with a ‘Theory of Mind’ in humans (Premack and Woodruff, 1978). Finally, our findings fit with recent results from Western scrub jays (Dally, Emery and Clayton, 2006) and chimpanzees Pan troglodytes (Hare, Call, Agnetta and Tomasello, 2000; Hare, Call and Tomasello, 2001), supporting the assumption that similar socio-cognitive skills have evolved convergently in some mammals and birds (Emery and Clayton, 2004). Still, we have to bear in mind that none of these studies provide unequivocal evidence for mental attribution (Penn and Povinelli, 2007; Shettleworth, 2010). The current studies on ravens are one of the first to reject the possibility that individuals merely associate what they have seen (i.e. the presence/absence of given individuals) with a high or low chance of competitively retrieving food (see also Bräuer, Call and Tomasello, 2007). Yet, there is much room for interpretation (Lurz, 2011) and further studies are needed to clarify which cognitive mechanisms non-human animals use for differentiating between ‘knowers’ and ‘guessers’.
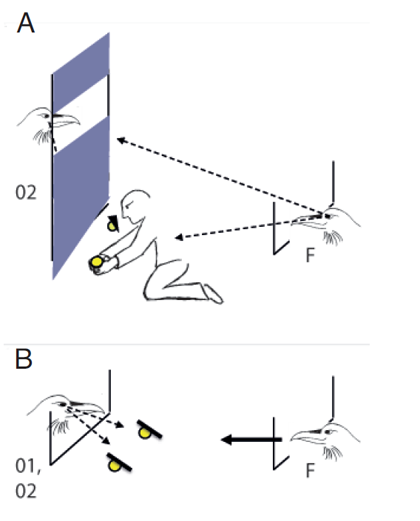
Figure 2. Sketch of the modified experimental set-up designed to dissociate the focal subject’s view from the view of its competitors. A. Potential competitors for pilfering (O1= bystander at cache 1, O2 = bystander at cache 2) are sitting on a perch. In the partially intact condition, a curtain is pulled down, blocking the competitors’ view towards the caches. However, the window in the upper part of the curtain ensures that the focal subject (F) can see the competitor at caching. B. At testing, the curtain is always pulled up and one of the competitors (O1 or O2) is sitting on the ground, next to the wire mesh. Arrows with broken lines symbolize gaze direction; arrow with full line symbolize head start of the focal subject.
How do ravens acquire their sophisticated knowledge about others at caching/pilfering? Given the numerous variables ravens have to consider for appropriate caching and successful pilfering, it not surprising to find strong evidence for learning, particularly during the first year of life (Bugnyar, Stöwe and Heinrich, 2007). However, making mistakes with food is costly: specifically when parents reduce the provisioning, young birds that loose their cached food to others may remain hungry. Interestingly, up to half of the caches made in the first months of life contain nonedible items only. These object caches are camouflaged in a similar way as food caches and readily elicit pilfer responses of other ravens (Bugnyar, Stöwe and Heinrich, 2007). Contrary to food caching, birds do not improve in object caching over time. Instead, they continue to show a high variation in when they make object caches and where they place them. For instance, birds could wait for a conspecific to approach and place an object cache directly in front of it or they could leave with an object and cache it far away/outside view from the other, as if to test the other’s response. Ravens also show this variation with unfamiliar human experimenters providing the birds with colored small objects, particularly during their first encounters (Bugnyar, Schwab, Schloegl, Kotrschal and Heinrich, 2007). When these human experimenters were instructed to behave towards the object caches in one of two ways, i.e. pilfer or just look at them, the ravens quickly learned to differentiate between ‘efficient’ and ‘nonefficient’ human pilferers of object caches. Importantly, they used this information only when caching food, i.e. they protected their food caches from ‘efficient’ pilferers but not from ‘non-efficient’ ones (Bugnyar, Schwab, Schloegl, Kotrschal and Heinrich, 2007). Playful caching of objects may thus represent a way of acquiring information about others, i.e. their behaviors at caching and/or pilfering, without the risk of losing precious food.
When taking a broad look at the ontogeny of cache protection and pilfering skills, two further points become apparent: first, it takes ravens a relatively long time to show full flexibility in their maneuvers, i.e. in appropriately using the learned information for deceiving others. Moreover, clear signs of concealment (hiding from view) come almost at same age in all subjects, hinting towards a developmental step (Bugnyar, Stöwe and Heinrich, 2007), possibly for controlling intentions. A similar age effect was found in the development of geometrical gaze following (Schloegl, Kotrschal and Bugnyar, 2007), indicating that the ability of tracking the other’s line of sight develops in close continuity with the ability of hiding from view. Secondly, ravens continue to play caching/pilfering with objects for years (Bugnyar, Schwab, Schloegl, Kotrschal and Heinrich, 2007). Possibly life in non-breeder groups with a high degree of fission-fusion dynamics require them to repeatedly learn about unfamiliar individuals and/or update information about hardly familiar individuals. In any case, it may indicate that ravens build up knowledge about others, often over years. The question then is how much they can use this information in daily life situations other than competition for caches?
Life in non-breeder groups: association patterns and social bonds
Anonymous aggregations or individualized societies? Ravens assembling at large food sources, like the moose carcass in the example above, could represent an anonymous crowd (Heinrich, 1988). However, birds of a given area regularly meet at nocturnal roosts, which they may use as information centers (Marzluff, Heinrich and Marzluff, 1996; Wright, Stone and Brown, 2003). Thus, it could be that (at least some) birds are quite familiar to one another. Preliminary support for this assumption comes from recent findings of subgroups with different foraging strategies (Dall and Wright, 2009), whereby ravens using the same roost tend to search for food together (Wright, Stone and Brown, 2003; but see Heinrich, Kaye, Knight and Schaumburg, 1994).
At our own study site in the Austrian Alps, we even find temporarily stable elements in non-breeder groups (Braun, Walsdorff, Fraser and Bugnyar, 2012; Braun and Bugnyar, in press). About one third of 200 individually-marked ravens develop a preference for particular foraging areas, using them almost on a daily basis. The majority of the marked population visits these areas from time to time, spending a few days up to months in the valley around the foraging sites. Together, this results in a moderate fluctuation in the composition of the foraging groups across months. Still, the non-breeders show a high degree of fission-fusion dynamics over the day, forming large groups for foraging and roosting but small groups of different composition for the rest of the day (Braun, Walsdorff, Fraser and Bugnyar, 2012).

Figure 3. An illustration of ravens engaging in preening, which is an analogue to grooming in primates. Photo by C. Schloegl
Specifically in these small groups, ravens engage in a range of socio-positive behaviors. They may share and offer food or, in a playful manner, show and offer non-food items (Pika and Bugnyar, 2011); they may sit in close contact (within reach of the other’s beak) and engage in reciprocal allopreening (analogue to grooming in primates) with particular individuals over extended time periods (Figure 3). This fits with reports from captive colonies (Lorenz, 1935; Gwinner, 1964; Heinrich, 1999) and suggests that ravens form affiliate relationships or social bonds. Importantly, such social bonds are not restricted to reproduction (mated pairs) but can be found in all age classes (juveniles in their first year, subadult and adults), between and within sexes and, to some extent, also between siblings of the same clutch (Fraser and Bugnyar, 2010a; Loretto, Fraser and Bugnyar, 2012; Braun and Bugnyar, in press).
Role of social bonds.
Having affiliates or bonding partners may pay off in various ways. Notably, it increases the individuals’ access to food. Under field conditions, this is true for both partners, irrespective of their sex and age class (Braun and Bugnyar, in press). In experiments with captive birds, the presence of bonded individuals (siblings, friends) affects the time spent in exploring new objects (Stöwe et al., 2006), the amount of attention paid to others (Scheid, Range and Bugnyar, 2007) and the likelihood of social learning (Schwab, Bugnyar, Schloegl and Kotrschal, 2008). Interestingly, ravens do not have many bonds simultaneously but focus on one up to a few partners at a time. However, bonding intensity and partners may change across seasons and years (Braun and Bugnyar, in press). Birds spending several years in non-breeder groups may thus end up with a decent number of affiliated individuals. Recent studies on captive ravens indicate excellent memory not only for former group members but also for the relationship valence they had with those individuals. Birds that were kept in one social group for two years responded differently to playbacks of calls from former affiliates and non-affiliates three years later (Boeckle and Bugnyar, 2012).
How comparable are avian social bonds to the primate/mammalian concept of bonds? Corvid social bonds reflect the content, quality and pattern of interactions over time (Emery, Seed, von Bayern and Clayton, 2007), fitting Hinde’s (1976) definition of social relationship. Following the theoretical framework of Cords and Aureli (2000), individual variation in relationship quality may be characterized by three main components: value, compatibility, and security. Interestingly, we found three main components describing the relationship quality of ravens (Fraser and Bugnyar, 2010a). Notably, these components were almost identical to those found in chimpanzees (Fraser, Stahl and Aureli, 2008) and, accordingly, they were labeled as value, compatibility and security. Kinship and sex combination had different effects on the components: kin relations were of high value, whereas male-male and male-female relations were more stable and secure than female-female relations, at least in this group of captive birds (Fraser and Bugnyar, 2010a).
As in primates (Fraser, Stahl and Aureli, 2008; Fraser, Koski, Wittig and Aureli, 2009), the components of relationship quality are a good predictor for the ravens’ behavior during and after conflicts (Fraser and Bugnyar, 2010b, 2011, 2012). Birds may provide help in ongoing conflicts by joining others in fights and/or chases, whereby they tend to support their kin and those who preened them and helped them before (Fraser and Bugnyar, 2012). Ravens may also show post-conflict behavior when they were engaged in the conflict themselves but also when they were just bystanders.
Reconciliation between former opponents is expected to occur when those individuals share a valuable relationship Aureli and de Waal, 2000). Ravens do show this pattern (Fraser and Bugnyar, 2011) but not in all studies (Fraser and Bugnyar, 2010b), probably due to the rare occurrence of conflicts between close affiliates. Much more often than reconciliation, bystander affiliation can be observed. Notably, if victims of aggression share a valuable relationship with one of the bystanders, they may seek its affiliation or actively get approached by that bystander for affiliate contacts (Fraser and Bugnyar, 2010b; see Seed, Clayton and Emery, 2007 for a similar findings in rooks, Corvus frugilegus). By initiating bystander affiliation, victims may try to protect themselves from any renewed aggression. In contrast, when a third party initiates affiliation, this usually follows a severe conflict (Fraser and Bugnyar, 2010b). Importantly, in those cases the value of the relationship between the bystander and the victim of aggression is higher than the value of the relationship between the bystander and the aggressor, indicating that the affiliate behavior towards the victim may serve as consolation rather than as a form of mediated reconciliation compare Wittig, Crockford, Wikberg, Seyfarth, and Cheney, 2007; Fraser, Koski, Wittig and Aureli, 2009). Furthermore, we found no signs of incompatibility or insecurity in the relationship between the bystander and the victim of aggression, suggesting that there is no need for the bystander to protect itself from redirection of conflicts by the victim (compare Koski and Sterck, 2007).
Synopsis
Recent research on wild and captive ravens allows us to identify two lines of potential driving forces for evolution of cognitive skills in this bird species, (i) competition over food caches and (ii) forming and maintaining social bonds. The former may go together with improved inhibition skills (control of intentions) and the deceptive use of social knowledge e.g. when/from whom to hide). Moreover, competition for hidden food likely results in elements of perspective taking (in the sense of understanding visual barriers, projecting other’s line of site) and, possibly, attribution skills (in the sense of remembering who could and could not see the caching). Although our understanding of the social structure of avian groups with high degrees of fission-fusion dynamics and seasonal patterns in group composition is still rudimentary, it appears safe to say that raven non-breeder groups are structured by different forms of social relationships.
Notably, birds form social bonds not only for reproduction; they can be found in several age classes, probably because bonded individuals serve as cooperation partners for gaining status and/or access to resources. The quality of raven social relationships resembles that reported for chimpanzees and, similar to mammals, the value of their bonds becomes apparent in alliance formation and conflict management. Hence, similar socio-cognitive skills may evolve independently of phylogeny in systems with a given degree of social complexity.
Relating these findings back to the arguments raised in the introduction, we may end up with the problem of how to classify species like ravens in the social spectrum? This question seems critical for any comparisons involving highly mobile species like birds that may spend time in smaller and larger groups depending on season and/or their stage in life history. Clearly, using breeding system (e.g. Iwaniuk and Arnold, 2004) or mean foraging group size (e.g. Emery, 2006) as proxi for complexity does not cover the full picture.
Taking aspects of relationship quality such as a long-term pair bond into account (e.g. Dunbar and Shultz, 2007; Emery, Seed, von Bayern and Clayton, 2007) has been a step forward; understanding how many individuals are dealt with on a personal basis and what type of relationships are important in what context and/or period of life seem to be the next logical steps. Characterizing the ‘complexity’ of social systems in more than one dimension may thus be a possible solution to our problem.
Research on social cognition has already gained much from broadening the focus to species outside of primates (e.g. McComb, Moss, Sayialel and Baker, 2000; Holekamp, Sakai and Lundrigan, 2007), as this allows testing assumptions independently of phylogeny. The value of including an even broader range of species into the picture, notably those classified as moderately social, is that it may allow us to refine our current models and may help to specify what aspects of social life select for which type of socio-cognitive skills.
References
Andersson, M. & Krebs, J.R. (1978). On the evolution of hoarding behaviour. Animal Behaviour, 26, 707-711. doi.org/10.1016/0003-3472(78)90137-9
Amici, F., Aureli, F. & Call, J. (2008). Fission-fusion dynamics, behavioural flexibility, and inhibitory control in primates. Current Biology, 18, 1415-1419. doi.org/10.1016/j.cub.2008.08.020 PMid:18804375
Aureli, F. & de Waal, F.B.M. (2000). Natural conflict resolution. Berkeley: University of California Press.
Aureli, F., Schaffner, C.M., Boesch, C., Bearder, S.K., Call, J., Chapman, C.A., Connor, R., Di Fiore, A., Dunbar, R.I.M., Henzi, S.P., Holekamp, K., Korstjens, A.H., Layton, R., Lee, P., Lehmann, J., Manson, J.H., Ramos-Fernandez, G., Strier, K.B. & van Schaik, C.P. (2008). Fission-fusion dynamics, new research frameworks. Current Anthropology, 49, 627-654. doi.org/10.1086/586708
Barrett, L., Henzi, S.P. & Dunbar, R.I.M. (2003). Primate cognition: from ‘what now’ to ‘what if’. Trends in Cognitive Sciences, 7, 494-497. doi.org/10.1016/j.tics.2003.09.005 PMid:14585446
Bednekoff, P.A. & Balda, R.P. (1996a). Social caching and observational spatial memory in Pinyon jays. Behaviour, 133, 807-826. doi.org/10.1163/156853996X00251
Bednekoff, P.A. & Balda, R.P. (1996b). Observational spatial memory in Clark’s nutcrackers and Mexican jays. Animal Behaviour, 52, 833-839. doi.org/10.1006/anbe.1996.0228
Boarman, W.I. & Heinrich, B. (1999). Common raven. The Birds of North America, 476, 1-31.
Boeckle, M. & Bugnyar, T. (2012). Adult ravens remember relationship valence they had with others as non-breeders. Current Biology, 22, 801-806. doi.org/10.1016/j.cub.2012.03.023 PMid:22521788 PMCid:3348500
Bond, A.B., Kamil, A.C. & Balda, R.P. (2003). Social complexity and transitive interference in corvids. Animal Behaviour, 65, 479-487. doi.org/10.1006/anbe.2003.2101
Bond, A.B, Wei, C.A. & Kamil, A.C. (2010). Cognitive representation in transitive inference: a comparison of four corvid species. Behavioural Processes, 85, 283-292. doi.org/10.1016/j.beproc.2010.08.003 PMid:20708664 PMCid:2975857
Braun, A. & Bugnyar, T. (2012). Social bonds and rank acquisition in raven nonbreeder aggregations. Animal Behaviour, 84, 1507-1515. doi.org/10.1016/j.anbehav.2012.09.024 PMid:23264693 PMCid:3518779
Braun, A., Walsdorff, T., Fraser, O.N. & Bugnyar, T. (2012). Socialized sub-groups in a temporarily-stable raven flock? Journal of Ornithology, 153, 97-104. doi.org/10.1007/s10336-011-0810-2
Bräuer, J., Call, J. & Tomasello, M. (2007). Chimpanzees really know what others can see in a competitive situation. Animal Cognition, 10, 439-448. doi.org/10.1007/s10071-007-0088-1 PMid:17426993
Bugnyar, T. (2011). Knowledge attribution in ravens: others’ viewpoints matter. Proceedings Royal Society London Series B, 278, 634-640. doi.org/10.1098/rspb.2010.1514 PMid:20826480 PMCid:3025684
Bugnyar, T. & Heinrich, B. (2005). Ravens, Corvus corax, differentiate between knowledgeable and ignorant competitors. Proceedings Royal Society London Series B, 272, 1641-1646. doi.org/10.1098/rspb.2005.3144 PMid:16087417 PMCid:1559847
Bugnyar, T. & Heinrich, B. (2006). Pilfering ravens, Corvus corax, adjust their behaviour to social context and identity of competitors. Animal Cognition, 9, 369-376. doi.org/10.1007/s10071-006-0035-6 PMid:16909235
Bugnyar, T. & Kotrschal, K. (2002a). Scrounging tactics in free-ranging ravens. Ethology, 108, 993-1009 doi.org/10.1046/j.1439-0310.2002.00832.x
Bugnyar, T. & Kotrschal, K. (2002b). Observational learning and the raiding of food caches in ravens, Corvus corax: Is it ‘tactical’ deception? Animal Behaviour, 64, 185-195 doi.org/10.1006/anbe.2002.3056
Bugnyar, T. & Kotrschal K. 2004 Leading a conspecific away from food in ravens, Corvus corax? Animal Cognition, 7, 69-76. doi.org/10.1007/s10071-003-0189-4 PMid:15069605
Bugnyar, T., Stöwe, M. & Heinrich, B. (2004). Ravens, Corvus corax, follow gaze direction of humans around obstacles. Proceedings Royal Society London Series B, 271, 1331-1336 doi.org/10.1098/rspb.2004.2738 PMid:15306330 PMCid:1691735
Bugnyar, T., Stöwe, M. & Heinrich, B. (2007). The ontogeny of caching in ravens, Corvus corax. Animal Behaviour, 74, 757-767. doi.org/10.1016/j.anbehav.2006.08.019
Bugnyar, T., Schwab, C., Schloegl, C., Kotrschal, K. & Heinrich, B. (2007). Ravens judge competitors through experience with play caching. Current Biology, 17, 1804-1808. doi.org/10.1016/j.cub.2007.09.048 PMid:17949980
Cameron, E.Z., Setsaas, T.H. & Linklater, W.L. (2009). Social bonds between unrelated females increase reproductive success in feral horses. Proceedings of the National Academy of Sciences of the United States of America, 106, 13850-13853. doi.org/10.1073/pnas.0900639106 PMid:19667179 PMCid:2728983
Cheney, D.L. & Seyfarth, R.M. (1990). How monkeys see the world. Chicago: University of Chicago Press. PMCid:175992
Cheney, D.L. & Seyfarth, R.M. (2007). Baboon Metaphysics. The Evolution of a Social Mind. Chicago: University of Chicago Press.
Clary, D. & Kelly, D.M. (2011). Cache protection strategies of a non-social food-caching corvid, Clark’s nutcracker (Nucifraga columbiana). Animal Cognition, 14, 735-744. doi.org/10.1007/s10071-011-0408-3 PMid:21538135
Clayton, N.S. & Emery, N.J. (2004). Cache robbing. In: Bekoff, M. & Goodall, J. (eds.) Encycopedia of Animal Behaviour (pp. 251-252). Westport, CT: Greenwood Publishing Group.
Clayton, N.S., Dally, J.M. & Emery, N.J. (2007). Social cognition by food caching corvids. The western scrub jay as natural psychologist. Philosophical Transactions of the Royal Society of London, Series B, 362, 507-522. doi.org/10.1098/rstb.2006.1992 PMid:17309867 PMCid:2346514
Cords, M. & Aureli, F. (2000). Reconciliation and relationship qualities. In: Aureli, F. & de Waal, F.B.M. (eds.) Natural Conflict Resolution (pp. 177-198). Berkeley: University of California Press.
Dall, S.R.X. & Wright, J. (2009). Rich pickings near large communal roosts favor ‘gang’ foraging by juvenile common ravens, Corvus corax. PLoS ONE, 4, e4530. doi.org/10.1371/journal.pone.0004530 PMid:19240813 PMCid:2646834
Dally, J.M., Emery, N.J. & Clayton, N.S. (2005). Cache protection strategies by Western scrub-jays (Aphelocoma californica): Implications for social cognition. Animal Behaviour, 70, 1251-1263. doi.org/10.1016/j.anbehav.2005.02.009
Dally, J.M, Emery, N.J. & Clayton, N.S. (2006). Food-caching scrub-jays keep track of who was watching when. Science, 312, 1662-1665. doi.org/10.1126/science.1126539 PMid:16709747
de Kort, S.R., Tebbich, S., Dally, J.M., Emery, N.J. & Clayton, N.S. (2006). The comparative cognition of caching. In: Zentall, T.R. & Wasserman, E. (eds.) Comparative Cognition (pp. 602-618). New York: Oxford University Press.
de Waal, F.B.M. & Tyack, P.L. (2003). Animal social complexity. Intelligence, culture, and individualized societies. Cambridge, MA: Harvard University Press.
Dunbar, R.I.M. (1992). Neocortex size as a constraint on group size in primates. Journal of Human Evolution, 20, 469 – 493. doi.org/10.1016/0047-2484(92)90081-J
Dunbar, R.I.M. (1998). The social brain hypothesis. Evolutionary Anthropology, 6, 178 – 190. doi.org/10.1002/(SICI)1520-6505(1998)6:5 3.0.CO;2-8 doi.org/10.1002/(SICI)1520-6505(1998)6:5 3.3.CO;2-P
Dunbar, R.I.M. & Shultz, S. (2007). Evolution in the social brain. Science, 317, 3144-3147. doi.org/10.1126/science.1145463 PMid:17823343
Emery, N.J. (2006). Cognitive ornithology: the evolution of avian intelligence. Philosophical Transactions of the Royal Society of London, Series B, 361, 23-43. doi.org/10.1098/rstb.2005.1736 PMid:16553307 PMCid:1626540
Emery, N.J. & Clayton, N.S. (2001). Effects of experience and social context on prospective caching strategies by scrub jays. Nature, 414, 443-446. doi.org/10.1038/35106560 PMid:11719804
Emery, N.J. & Clayton, N.S. (2004). The mentality of crows: convergent evolution of intelligence in corvids and apes. Science, 306, 1903-1907. doi.org/10.1126/science.1098410 PMid:15591194
Emery, N.J., Seed, A.M., von Bayern, A.M.P. & Clayton, N.S. 2007. Cognitive adaptions of social bonding in birds. Philosophical Transactions of the Royal Society of London, Series B, 362, 489-505. doi.org/10.1098/rstb.2006.1991 PMid:17255008 PMCid:2346513
Fraser, O.N. & Bugnyar, T. (2010a). The quality of social relationships in ravens. Animal Behaviour, 79, 927-933. doi.org/10.1016/j.anbehav.2010.01.008
Fraser, O.N. & Bugnyar, T. (2010b). Do ravens show consolation? Responses to distressed other. PLoS One, 5, e10605. doi.org/10.1371/journal.pone.0010605 PMid:20485685 PMCid:2868892
Fraser, O.N. & Bugnyar, T. (2011). Ravens reconcile after conflicts with valuable partners. PLoS One, 6, e18118. doi.org/10.1371/journal.pone.0018118 PMid:21464962 PMCid:3064662
Fraser, O.N. & Bugnyar, T. (2012). Reciprocity of agonistic support in ravens. Animal Behaviour, 83, 171-177 doi.org/10.1016/j.anbehav.2011.10.023 PMid:22298910 PMCid:3255075
Fraser, O.N., Stahl, D. & Aureli, F. (2008). Stress reduction through consolation in chimpanzees. Proceedings of the National Academy of Sciences of the United States of America, 105, 8557-8562. doi.org/10.1073/pnas.0804141105 PMid:18559863 PMCid:2438392
Fraser, O.N., Koski, S.E., Wittig, R.M. & Aureli, F. (2009). Why are bystanders friendly to recipients of aggression? Communicative & Integrative Biology, 2, 285-291. doi.org/10.4161/cib.2.3.8718 PMid:19641753 PMCid:2717543
Giraldeau, L.-A. & Caraco, T. (2000). Social Foraging Theory. Princeton, New Jersey: Princeton University Press.
Gwinner, E. (1964). Untersuchungen über das Ausdrucks- und Sozialverhalten des Kolkraben (Corvus corax corax L.). Zeitschrift für Tierpsychologie, 21, 657-748. doi.org/10.1111/j.1439-0310.1964.tb01212.x
Hare, B., Call. J., Agnetta, B. & Tomasello, M. (2000). Chimpanzees know what conspecifics do and do not see. Animal Behaviour, 59, 771-785. doi.org/10.1006/anbe.1999.1377 PMid:10792932
Hare, B., Call. J. & Tomasello, M. (2001). Do chimpanzees know what conspecifics know? Animal Behaviour, 61, 139-151. doi.org/10.1006/anbe.2000.1518 PMid:11170704
Harriman, A.E. & Berger, R.H. (1986). Olfactory acuity in the common raven (Corvus corax). Physiological Behavior, 36, 257-262. doi.org/10.1016/0031-9384(86)90013-2
Heinrich, B. (1989). Ravens in winter. New York: Simon & Schuster.
Heinrich, B. (1999). Mind of the raven. New York Harper-Collins.
Heinrich, B. & Marzluff, J.M. (1991). Do common ravens yell because they want to attract others? Behavioural Ecology and Sociobiology, 28, 13-21. doi.org/10.1007/BF00172134
Heinrich, B. & Pepper, J.R. (1998). Influence of competitors on caching behaviour in common ravens, Corvus corax. Animal Behaviour, 56, 1083-1090. doi.org/10.1006/anbe.1998.0906 PMid:9819322
Heinrich, B., Kaye, D., Knight, T. & Schaumburg, K. (1994). Dispersal and association among common ravens. Condor, 96, 545-551. doi.org/10.2307/1369334
Hinde, R.A. (1976). Interactions, relationships and social structure. Man, 11, 1-17. doi.org/10.2307/2800384
Holekamp, K.E., Sakai, S.T. & Lundrigan, B.L. (2007). Social intelligence in the spotted hyena (Crocuta crocuta). Philosophical Transactions of the Royal Society of London, Series B, 362, 523-538. doi.org/10.1098/rstb.2006.1993 PMid:17289649 PMCid:2346515
Humphrey, N.K. (1976). The social function of intellect. In: Bateson, P. & Hinde, R.A. (eds) Growing Points in Ethology (pp. 303-321). Cambridge: Cambridge University Press.
Iwaniuk, A.N. & Arnold, K.E. (2004). Is cooperative breeding associated with bigger brains? A comparative test in the corvida (Passeriformes). Ethology, 110, 203-220. doi.org/10.1111/j.1439-0310.2003.00957.x
Jolly, A. (1966). Lemur social behavior and primate intelligence. Science, 153, 501-507. doi.org/10.1126/science.153.3735.501 PMid:5938775
Källander, H. (2007). Food hoarding and use of stored food by rooks Corvus frugilegus. Bird Study, 54, 192-198. doi.org/10.1080/00063650709461475
Koski, S.E. & Sterck, E.H.M. (2007). Triadic postconflict affiliation in captive chimpanzees: does consolation console? Animal Behaviour, 73, 133-142. doi.org/10.1016/j.anbehav.2006.04.009
Lorenz, K. Z. (1935). Der Kumpan in der Umwelt des Vogels. Journal für Ornithologie, 83, 137-215 and 289-413. doi.org/10.1007/BF01905572 doi.org/10.1007/BF01905355
Loretto, M.-C., Fraser, O.N. & Bugnyar, T. (2012). Ontogeny of social relations and coalition formation in common ravens (Corvus corax). International Journal of Comparative Psychology, in press.
Lurz, R. (2011). Belief attribution in animals: on how to move forward conceptually and empirically. Review of Philosophy and Psychology, 22, 19-59. doi.org/10.1007/s13164-010-0042-z
McComb, K., Moss, C., Durant, S., Baker, L. & Sayialek, S. (2001). Matriarchs act as repositories of social knowledge in African elephants. Science, 292, 491-494. doi.org/10.1126/science.1057895 PMid:11313492
McComb, K., Moss, C., Sayialel, S. & Baker, L. (2000). Unusually extensive networks of vocal recognition in African elephants. Animal Behaviour, 59, 1103-1109. doi.org/10.1006/anbe.2000.1406 PMid:10877888
Mann, J., Connor, R.C., Tyack, P. & Whitehead, H. (2000). Cetacean societies: field studies on dolphins and whales. Chicago: University of Chicago Press.
Marzluff, J.M. & Angell, T. (2005). In the company of crows and ravens. New Haven: Yale University Press.
Marzluff, J.M. & Balda, R.P. (1992). The pinyon jay. Behavioral ecology of a colonial and cooperative corvid. San Diego: Academic Press.
Marzluff, J.M. & Heinrich, B. (1991). Foraging by common ravens in the presence and absence of territory holders – an experimental analysis of social foraging. Animal Behaviour, 42, 755-770. doi.org/10.1016/S0003-3472(05)80121-6
Marzluff, J.M., Heinrich, B. & Marzluff, C.S. (1996). Roosts are mobile information centers. Animal Behaviour, 51, 89-103. doi.org/10.1006/anbe.1996.0008
Milton, K. (1988). Foraging behaviour and the evolution of primate intelligence. In: Byrne, R.W. & Whiten, A. (eds.) Machiavellian intelligence. Social expertise and the evolution of intellect in monkeys, apes, and humans (pp. 285-305). New York: Oxford University Press.
Moll, H. & Tomasello, M. (2007). Cooperation and human cognition: the Vygotskian intelligence hypothesis. Philosophical Transactions of the Royal Society of London, Series B, 362, 639-648. doi.org/10.1098/rstb.2006.2000 PMid:17296598 PMCid:2346522
Parker, S.T. & Gibson, K.R. (1977). Object manipulation, tool use and sensorimotor intelligence as feeding adaptations in Cebus monkeys and great apes. Journal of Human Evolution, 6, 623-641. doi.org/10.1016/S0047-2484(77)80135-8
Penn, D.C. & Povinelli, D.J. (2007). On the lack of evidence that non-human animals possess anything remotely resembling a ‘theory of mind’. Philosophical Transactions of the Royal Society of London, Series B, 362, 731-744. doi.org/10.1098/rstb.2006.2023 PMid:17264056 PMCid:2346530
Pérez-Barbería, F.J., Shultz, S. & Dunbar, R.I.M. (2007). Evidence for coevolution of sociality and relative brain size in three orders of mammals. Evolution, 61, 2811-2821. doi.org/10.1111/j.1558-5646.2007.00229.x PMid:17908248
Pika, S. & Bugnyar, T. (2011). The use of referential gestures in ravens (Corvus corax) in the wild. Nature Communications, 2, 560. doi.org/10.1038/ncomms1567 PMid:22127056
Povinelli, D.J. & Vonk, J. (2003). Chimpanzee minds: suspiciously human? Trends in Cognitive Sciences, 7, 157-160. doi.org/10.1016/S1364-6613(03)00053-6
Premack, D. & Woodruff, G. (1978). Does the chimpanzee have a theory of mind? Behavioral and Brain Sciences, 1, 515-526. doi.org/10.1017/S0140525X00076512
Ratcliffe, D. (1997). The Raven. A Natural History in Britain and Ireland. London: T. & A.D. Poyser. Scheid, C., Range, F. & Bugnyar, T. (2007). When, what, and whom to watch? Quantitive measures of attention to conspecifics in ravens (Corvus corax) and jackdaws (Corvus monedula). Journal of Comparative Psychology, 121, 380-386. doi.org/10.1037/0735-7036.121.4.380 PMid:18085921
Schloegl, C., Kotrschal, K. & Bugnyar, T. 2007. Gaze following in Common ravens (Corvus corax): Ontogeny and habituation. Animal Behaviour, 74, 769-778. doi.org/10.1016/j.anbehav.2006.08.017
Schloegl, C., Kotrschal, K. & Bugnyar, T. (2008). Do common ravens (Corvus corax) rely on human or conspecific gaze cues to detect hidden food? Animal Cognition, 11, 231-241. doi.org/10.1007/s10071-007-0105-4 PMid:17762942
Schwab, C., Bugnyar, T., Schlögl, C. & Kotrschal, K. (2008). Enhanced social learning between siblings in common ravens (Corvus corax). Animal Behaviour, 75, 501-508. doi.org/10.1016/j.anbehav.2007.06.006
Seed, A.M., Clayton, N.S. & Emery, N.J. (2007). Postconflict third-party affiliation in rooks, Corvus frugilegus. Current Biology, 17, 1-7. doi.org/10.1016/j.cub.2006.11.025 PMid:17240341
Shettleworth, S.J. (2010). Clever animals and killjoy explanations in comparatve psychology. Trends in Cognitive Sciences, 14, 477-481. doi.org/10.1016/j.tics.2010.07.002 PMid:20685155
Silk, J.B., Alberts, S.C. & Altmann, J. (2003). Social bonds of female baboons enhance infant survival. Science, 302, 1231-1234. doi.org/10.1126/science.1088580 PMid:14615543
Stöwe, M., Bugnyar, T., Loretto, M.-C., Schlögl, C., Range, F. & Kotrschal, K. (2006). Novel object exploration in ravens (Corvus corax): effects of social relationships. Behavioral Processes, 73, 68-75. doi.org/10.1016/j.beproc.2006.03.015 PMid:16682154
Van der Vaart, E., Verbrugge, R. & Hemelrijk, C.K. (2012). Corvid re-caching without a ‘Theory of Mind’: a model. PLoS ONE, 7, e32904. doi.org/10.1371/journal.pone.0032904 PMid:22396799 PMCid:3291480
Vander Waal, S.B. & Jenkins, S.H. (2003). Reciprocal pilferage and the evolution of food hoarding behavior. Behavioral Ecology, 14, 656-667. doi.org/10.1093/beheco/arg064
Whiten, A. & Byrne, R.W. (1988). The Machiavellian intelligence hypotheses. In: Byrne, R.W. & Whiten, A. (eds.) Machiavellian intelligence: social complexity and the evolution of intellect in monkeys, apes and humans (pp. 1-9). Oxford: Oxford University Press.
Wittig, R.M., Crockford, C., Wikberg, E., Seyfarth, R.M. & Cheney, D.L. (2007). Kin-mediated reconciliation substitutes for direct reconciliation in female baboons. Proceedings of the Royal Society of London,Series B, 274, 1109-1115 doi.org/10.1098/rspb.2006.0203 PMid:17301022 PMCid:2124468
Wright, J., Stone, R.E., and Brown, N. (2003). Communal roosts as structured information centres in the raven, Corvus corax. Journal of Animal Ecology, 72, 1003–1014. doi.org/10.1046/j.1365-2656.2003.00771.x
How to Reference This Article:
Bugnyar, T. (2013). Social cognition in ravens. Comparative Cognition & Behavior Reviews, 8, 1-12. Retrieved from https://comparative-cognition-and-behavior-reviews.org/index.html doi:10.3819/ccbr.2013.80001
